Lab Grown Diamonds
vs Mined Diamonds
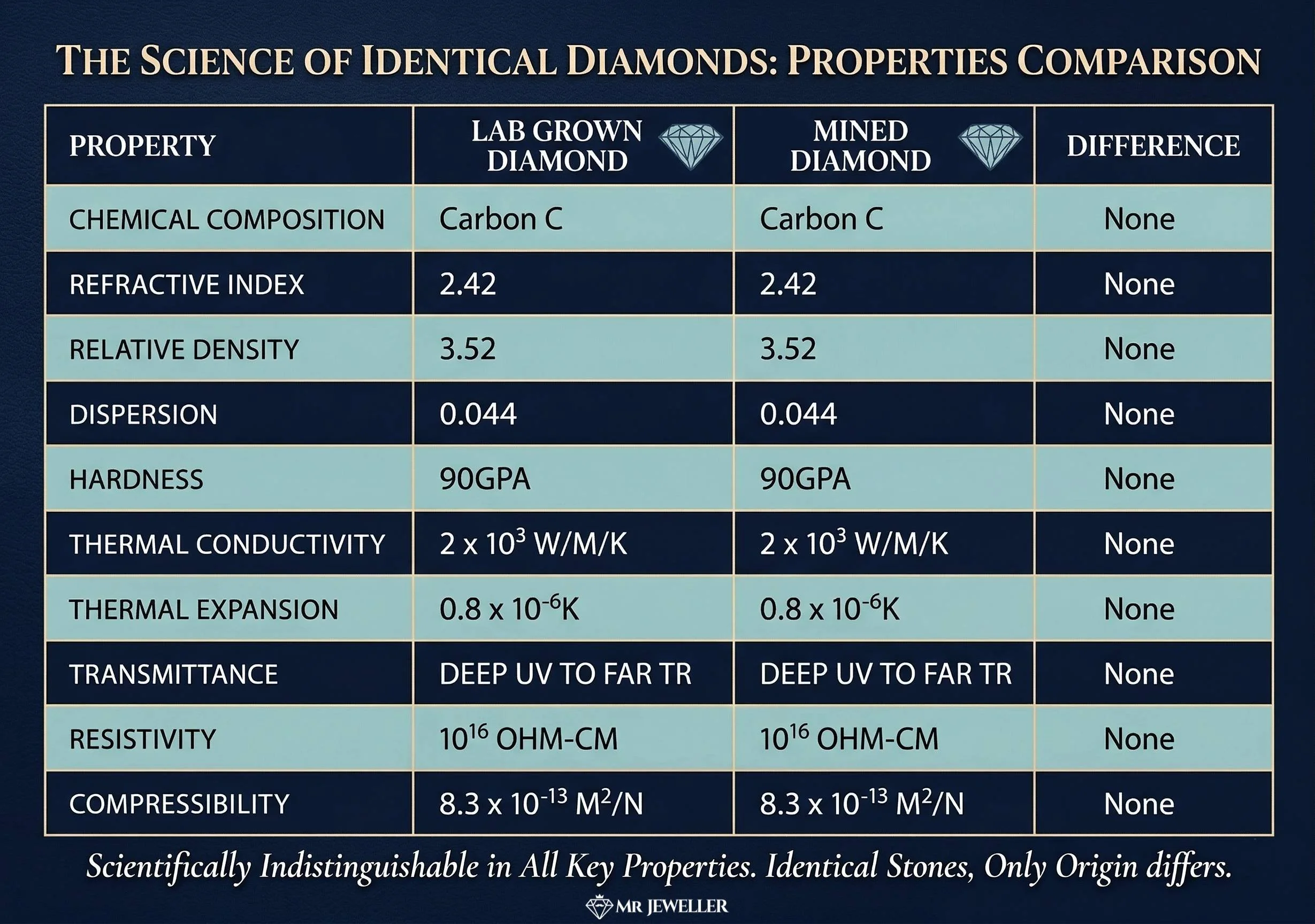
Lab-grown diamonds and natural mined diamonds both rank as a perfect 10 on the Mohs scale of hardness (which is 4x harder than a 9).
Scientifically, real diamonds created in a laboratory have the exact same physical and chemical properties as diamonds that are mined from the earth’s surface.
At Mr. Jeweller, we meet customers every day who are looking for a symbol: a perfect, enduring testament to their love, their achievement, or their commitment.
For over a century, the automatic response to that need has been simple: “I need a diamond.”
But today, that response is followed immediately by a difficult, confusing question: “Mined or Lab-Grown?”
We understand the confusion. You are making a significant financial and emotional investment. You’ve heard myths and marketing spin from all sides. You’re worried that lab-grown might mean "synthetic," "fake," or "imitation."
You’re worried you will sacrifice the eternal nature of the gift by choosing a modern approach.
At Mr. Jeweller, we specialize in lab-grown diamonds. We didn't make this choice lightly. We scrutinized the data, the science, and the results, and we realized that the prevailing story about mined diamonds vs. lab-grown diamonds is riddled with outdated concepts.
The single most important thing you need to know, the absolute prerequisite for your confidence, is this:
A Lab-Grown Diamond Is a Real Diamond. Period.
We are not "substituting." We are not selling a simulation. We are not offering a "close enough" alternative like cubic zirconia (CZ) or moissanite. Those are different materials with entirely different chemical compositions, densities, and optical properties.
A lab-grown diamond is a masterstroke of modern human engineering.
To fully grasp this, you need to understand that "diamond" is not a description of origin. It is a description of substance. Chemically, physically, and optically, they are identical.
We’ll explore the science and use three illustrations to prove to you that your eyes, your heart, and even a geologist’s tools cannot tell the difference, because there is no difference to tell.

Illustration 1: Ice Cubes
Consider the ice cube floating in your glass of water. Where did it come from?
It was almost certainly created by the freezer attached to your refrigerator. It is clean, clear, predictable, and available on demand. Now, imagine a specialist harvesting a chunk of ice from the heart of a 10,000-year-old Arctic glacier.
The glacier ice was obtained more naturally. It required an expedition, a dangerous journey, heavy equipment, and a vast ecosystem to form over millennia. The freezer ice was created in two hours by electricity and a simple cooling loop.
But when you melt them down and analyze the water molecule, you will find H2O. Period. No spectrometer in the world can analyze the melted freezer water and say, “Ah, this isn't real water; it lacks the geological journey.” It’s water. One was obtained through nature's slow, unpredictable method; the other was obtained through human ingenuity replicating that same method in a clean environment.
At Mr. Jeweller, we sell the clean, pure, ethically obtained "freezer ice" of the diamond world. It’s indistinguishable from glacier ice when it matters.
How Human Ingenuity Mimics Nature’s Process
Geologically, diamonds form 100 miles below the earth's surface when pure carbon is subjected to pressures of up to 60,000 atmospheres and temperatures of nearly 2,000 degrees Fahrenheit. It is a slow, messy, brutal process that takes thousands of years. Mined diamonds are simply the stones that were forced near the earth’s surface by deep volcanic eruptions.
Lab-grown diamond producers didn’t invent a new gemstone; they invented two brilliant ways to edit that arduous journey down to a few months.
Process 1: High Pressure High Temperature (HPHT)
This process literally replicates the earth’s environment. Engineers place a tiny diamond "seed" (a minuscule fragment of diamond) in a specialized press. They surrounding the seed with pure carbon and subject it to the identical, staggering pressure and heat of the earth's mantle. Under these conditions, the carbon dissolves and crystallizes around the seed, growing a gem-quality diamond atom by single carbon atom.
Process 2: Chemical Vapor Deposition (CVD)
If HPHT is the sledgehammer approach, CVD is the scalpel. This process takes a diamond seed and places it in a vacuum chamber. Engineers inject gases (usually methane and hydrogen) and ignite them into a plasma state. At this point, the carbon atoms in the gas are released and raining down, depositing themselves onto the diamond seed, growing the stone layer by microscopic layer.
Both processes produce a result that is 100% pure carbon in an isometric-cubic crystal lattice structure. This structure is exactly what makes a diamond the hardest natural material on Earth. A lab-grown diamond scores a perfect 10 on the Mohs scale of hardness, making it a "real" diamond in durability and timelessness.

Illustration 2: The Greenhouse Rose
Imagine walking into a high-end flower shop to buy a bouquet of red roses. You pick a perfect, velvety stem with an intoxicating scent. Its petals are flawles, its color vibrant.
Now imagine asking the florist, "Where was this rose grown?"
One rose might have been harvested from a dusty garden, fighting insects, erratic rainfall, and unpredictable temperature shifts, developing "natural" inclusions like scarred petals or slightly weaker stems.
Another identical rose might have been grown in a highly technical, computerized greenhouse, where every variable (from the light spectrum to the nutrient blend) was optimized.
This greenhouse rose is perfect. It has the same DNA, the same scent molecules, the same cellular structure as the garden rose. It is exactly the same flower, just grown in a perfect environment.
You would never call the greenhouse rose a "fake rose" or an "imitation flower." You would call it a superior flower. You would call it a rose grown better.
Lab-grown diamonds are Mr. Jeweller’s greenhouse roses. We remove the dirt, the unpredictability, and the harsh elements of the natural garden of the earth’s mantle to provide you with a stone grown in optimized perfection.
Why Experts Can’t Tell (Without Special Tools)
The myth that “a trained eye can tell the difference” is just that—a myth.
Mined and lab-grown diamonds are both evaluated using the exact same four criteria established by the Gemological Institute of America (GIA): Cut, Color, Clarity, and Carat.
Mined diamonds are formed with impurities. The most common impurity is nitrogen, which gives some mined stones a slight yellow tint. A modern lab can grow Type IIa diamonds, which are defined by their almost absolute lack of nitrogen. Type IIa is the rarest and purest chemical category of diamond. Only 2% of mined diamonds qualify. Our lab-grown process produces Type IIa stones reliably.
If you gave a sample of our diamonds and a sample of high-quality mined diamonds to a group of expert gemologists equipped only with standard jewelers’ loupes, they would be unable to spot the difference between the mined diamond and the lab-grown diamond. They would grade both samples for Cut, Color, Clarity, and Carat using the same instruments and the same standards.
The only way to distinguish them is by sending them to a major gemological laboratory (like GIA or IGI) for advanced spectrographic analysis with expensive equipment that evaluates the length of time over which the diamond was formed.

Two Advantages of Lab-Grown Diamonds
Hopefully you’re convinced that lab-grown diamonds are identical to natural diamonds. But we also want to show you why they are better. The differences between mined and lab-grown diamonds are not found in the stone; they are found in the cost to the environment and the cost to your wallet.
The Environmental Difference: A Lighter Footprint
The story of traditional diamond mining is one of enormous scale. Industrial mining requires the blasting of open-pit or underground mines that are miles wide and deep. For every single one-carat gem-quality diamond recovered from a mine, engineers often must move over 200 tons of earth and rock. This process consumes vast amounts of diesel fuel and uses millions of gallons of water. It is an extractive industry that, by definition, requires habitat destruction and permanent change to the landscape.
Our lab-grown stones are created in localized facilities. We can proudly say our process has:
90%+ fewer carbon emissions.
Significantly less water usage (by an order of magnitude).
Zero land destruction or ecosystem displacement.
When you choose a lab-grown diamond, you are editing out environmental destruction from the symbol of your love.
The Price Difference: Intelligent Luxury
This is the difference that most customers find truly winsome. Lab-grown diamonds are significantly less expensive than mined diamonds of the same quality.
This is not because they are lower quality. It is because we have edited the entire supply chain.
Mined Diamond Chain: Mine > Sorter > Rough Dealer > Manufacturer > Polished Dealer > Jewellery Brand > Store > You. Every link adds time, cost, and logistics.
Mr. Jeweller Lab-Grown Chain: Lab Facility > Mr. Jeweller > You.
We have removed the vast logistics of mining, the international rough shipping, and the speculative markets. We pass those savings directly to you.
You can purchase lab-grown diamond that is significantly larger than a mined diamond for the same budget, and with superior Color and Clarity.

Illustration 3: Mined Gold vs Refined Gold
Imagine a nugget of raw gold mined from deep beneath the Earth’s surface. Now, imagine a piece of refined, 24k gold. Where did that refined gold come from? It might have been melted down from discarded vintage jewelry, recovered from complex aerospace components, or recycled from outdated electronics in a smelter’s furnace.
Nature’s furnace vs. the smelter’s furnace.
Here’s the main point: If both pieces of gold are refined to 24k purity (99.99% pure), they are chemically, physically, and optically identical. A scientist or jeweler, looking at two pure 24k gold bars, cannot distinguish their origins. They are the exact same substance (Element Au). Gold is gold.
It’s the same with a diamond.
A mined diamond is extracted from the Earth’s mantle. A lab-grown diamond is created in a facility using advanced technology (the “smelter” in this analogy). Yet, both are 100% pure carbon. They both possess the exact same interlocking crystal lattice structure and achieve the highest possible rating of 10 on the Mohs scale of hardness.
Gemological institutes like the IGI use the exact same standards (the 4Cs: Cut, Color, Clarity, and Carat) to grade both mined and lab-grown diamonds because they are identical. By understanding this, you are free to make a decision based on your personal preferences for cost, environmental impact, and ethics, knowing that the brilliance and durability of your diamond are absolute.

The Remarkable Purity of Lab-Grown Diamonds
To understand why lab-grown diamonds are often “purer” than the earth's most elite offerings, we have to look at the Diamond Typing System.
The Diamond Typing System
Diamonds are categorized into two main types based on the presence of Nitrogen, which is the most common "typo" found in a diamond’s chemical composition.
Type I: The “Common” Diamond (98% of Mined Stones)
Most diamonds found in nature are Type I. They contain nitrogen atoms.
Type Ia: The nitrogen is in clusters. This can give diamonds a yellowish tint because the clusters absorb blue light.
Type Ib: The nitrogen is scattered as single atoms. These are very rare in nature and often have an intense yellow or "canary" color.
Type II: The “Pure” Diamond (2% of Mined Stones)
Type II diamonds have no measurable nitrogen. They are the "cleanest" sentences ever written in the language of geology.
Type IIb: Contains boron, which makes the diamond blue (like the famous Hope Diamond).
Type IIa: The "Perfect" Diamond. No nitrogen, no boron. Just pure carbon.
Why Lab-Grown Wins the Purity Race
In nature, a diamond is formed in a "dirty" environment. Deep underground, carbon is surrounded by nitrogen, boron, and other elements. It’s almost impossible for a diamond to grow for a billion years without catching a few nitrogen atoms along the way.
At Mr. Jeweller, our lab-grown diamonds are almost exclusively Type IIa.
Here is why that matters:
The "Greenhouse" Effect: In a CVD (Chemical Vapor Deposition) chamber, we control the atmosphere with $100\%$ precision. We remove the nitrogen from the air before the "growth" begins.
Supernatural Brilliance: Because Type IIa diamonds have no nitrogen to absorb light, they are more optically transparent. They allow more light to pass through and disperse, resulting in that "whiter-than-white" look that characterizes the world's most famous (and expensive) crown jewels.
The "Elite" Comparison: In the mining world, Type IIa diamonds are legendary. The Cullinan (the largest gem-quality rough diamond ever found) and the Koh-i-Noor (part of the British Crown Jewels) are Type IIa.
The Mr. Jeweller Insight: When you buy a lab-grown diamond from us, you aren't just getting a real diamond. You’re getting a diamond that is chemically superior to 98% of what is pulled out of the ground. You are buying the same chemical purity as the stones worn by royalty.

Illustration 4: Modern Investment vs. Collector Scarcity
When the automobile was invented, it was a handcrafted marvel of gears and wood, incredibly expensive and difficult to maintain. Over a century later, a brand-new, top-of-the-line electric luxury vehicle can outperform that vintage car in every single measurable metric: speed, acceleration, safety, reliability, comfort, and efficiency. It is functionally superior.
Yet, a collector's classic car from 1920 will cost ten times more than the modern electric vehicle. Why? It isn’t because it is a "better" car in the way you buy a car (to drive). It is expensive strictly because of scarcity—there are only a few left in the world.
The diamond industry has built a pricing structure around that vintage collector car scarcity. But you aren't trying to collect an ancient geological sample. You are buying a symbol of brilliance.
A lab-grown diamond is the modern electric luxury vehicle: it is functionally perfect, functionally brilliant, functionally timeless, and costs less because we have finally figured out the modern, efficient way to make them. Why pay a premium for "collector scarcity" when you can buy the functional ideal of a diamond?
Choose Your Future
The word "synthetic" has been used to frighten diamond buyers. It conjures images of cheap plastics or fakes. But in chemistry, "synthetic" simply means "produced by synthesis; man-made." It is a word about origin, not quality. Synthetic sapphire, synthetic ruby, and synthetic diamonds are the chemical counterparts of their natural counterparts.
At Mr. Jeweller, we don’t use the word "synthetic" because we know it leads you to the wrong conclusion. We use "lab-grown" because it is the winsome, truthful term. It acknowledges the hand of human innovation without diminishing the enduring substance of the stone.
Your decision should be based on beauty, on values, and on love—not an outdated story. By choosing lab-grown from Mr. Jeweller, you are choosing:
A 100% Real Diamond: Chemically, physically, and optically identical, scoring a 10 on the Mohs hardness scale.
A Higher Standard: Stones that are almost exclusively Type IIa (purest carbon) and graded to the highest standards.
A Lighter Footprint: The choice that protects our planet.
Intelligent Value: More diamond, better quality, for significantly less investment.
Let us help you edit the story of your symbol. Your love deserves a diamond that is both a scientific miracle and a responsible choice. Visit Mr. Jeweller today, look at the two identical stones side-by-side, and ask yourself why you would ever pay more to make the choice that costs the planet the most.

